

| Uranium Symbol: U Atomic Number: 92 The best-known member of the f-block for sure. Since the Cold War, the name of Uranium has been enough to send a shiver down the spine of the public. In truth; uranium is radioactive, but with a half-life of 4.5 billion years, its chemical toxicity is a far more serious danger than its radioactivity. It is fissile, or at least some of it is, but most uranium is not. And shown here is depleted uranium; a form of uranium which has had most of the fissile form removed. Uranium is in fact a rather beautiful metal, with a colour bringing to mind the likes of copper, silver, and gold, and some of its ions, like UO3+, also form rather beautifully-coloured minerals. But that is to say.... look, but don't touch! |
| Bromine Symbol: Br Atomic Number: 35 Bromine's name comes from a Greek word meaning 'stench'; a characteristic of all of the halogens. But bromine is truly unique as it is one of only two; the other being mercury; elements liquid at room temperature. And bromine is a rather glorious black liquid with an even more glorious reddish vapour. Bromine is often extracted from bromide salts; with the Dead Sea in the Middle East having significant bromide deposits. |
| Antimony Symbol: Sb Atomic Number: 51 Antimony's name may have come from a Latin compound word meaning 'never alone'...a trait hardly unique to antimony. In the Middle Ages, antimony was a valued medicine for constipation, as the body would work vigrously to release the toxin... and often also released the antimony pill unaffected. For those of you somehow curious enough to try this out, it should be warned that death has resulted from the expunging capabilities of antimony. |
| Gold Symbol: Au Atomic Number: 79 The 'gold standard' of all of the metals. Gold is neither the only metal which occurs uncombined in nature nor the most common, but its sheer tendency to stand out likely made it one of the first metals discovered by people. Since then, gold has been a metal lived, fought, searched, longed, and died for. At the Serra Pelada in Brazil, in 1983, the largest-known gold nugget to date was found. It weighed 61 kg [134.5 lb], but only contained 52 kg of the element known in Latin as aurum. So it was only 85% worth its weight in gold. |
| Aluminium Symbol: Al Atomic Number: 13 Although not as mythologised or as valued as other metals, Aluminium is still a very sturdy metal with many practical uses. In nature, aluminium never occurs uncombined. It occurs mainly in the aluminium-rich bauxite, which is really a potpurri of various minerals, but to squeeze aluminium out of bauxite requires much energy. So a significant amount of aluminium is recycled. It is from bauxite that aluminium's downstairs 'cousin', gallium, #31, is also extracted from. Gallium is famous because it is solid at room temperature but melts slightly above that - it can melt in the hand. But it is destructive to the structure of aluminium products such that its transport by plane is limited by law. |
| Carbon Symbol: C Atomic Number: 6 In essence the backbone of life itself, carbon assumes a myriad of forms, from graphite, as can be seen here, to diamond to graphene and buckyball. And it is with atoms of itself, carbon, and other atoms, that carbon forms a near infinite array of different compounds. So many that carbon has a field of chemistry all to itself, organic chemistry. The hardness of carbon varies significantly between its forms; layers of graphene have weak bonds between each other and slip over easily. The layers in diamond are strongly bonded and regular, making diamond one of the hardest known materials in nature. Now that graphite can be turned into diamond in the lab, one can only hope the terrible conflicts over this beautiful mineral will cease. |




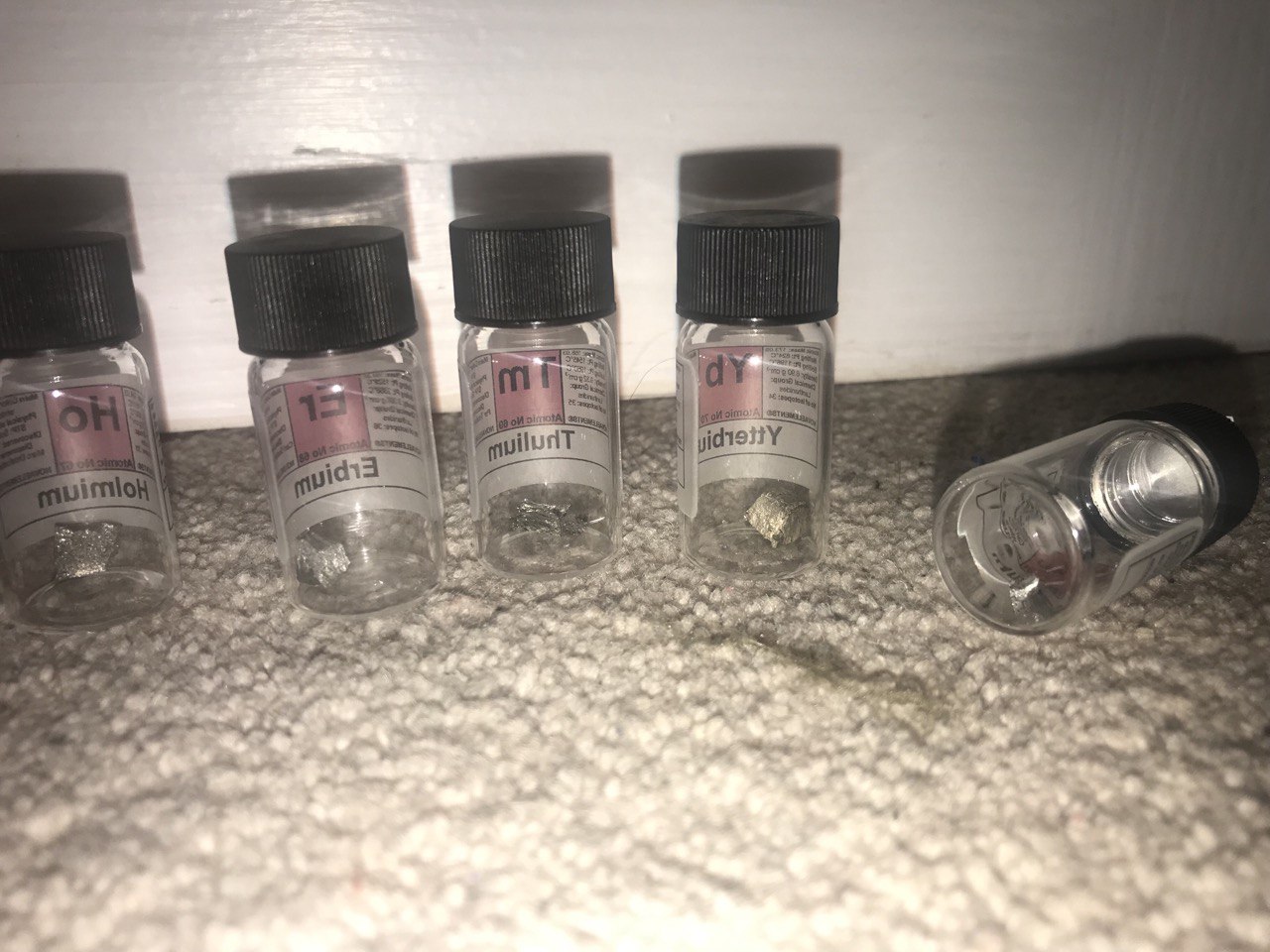

| The Lanthanides; the
rare-earths; the things the politicians love but the voters don't care
for The lanthanides, more often called the rare-earths, are such a politically savvy group of metals that I am pleased to now have a whole set of them; Lanthanum, Cerium, Praeseodymium, Neodymium, Promethium [only trace amounts], Samarium, Europium, Gadolinium, Terbium, Dysprosium, Holmium, Erbium, Thulium, Ytterbium and Lutetium [which some dispute whether it should be counted a lanthanide or not]. The 'rare-earths' are not truly rare. 'Earth' is an old term for most solid elements; they were once believed to be rare, but only because they all usually occur together because of chemical similarity. The most common lanthanide, Cerium, is more common than lead and nickel. The rarest with a stable isotope,Thulium, still manages to be more common than silver. Promethium is rarest of all; it is radioactive, occurring in nature either from the fission of Uranium or the decay of Europium, which is only slightly radioactive [and not all of it is, as far as we know]. In promethium's place here is a Europium glow powder. |
| In late 2019, one of the more exciting pastimes
of my life began, when I received the first twelve elements of my
element collection. Over the past - wow - nearly 7 years since then,
the household periodic table has ballooned in size to accomodate most
of its elements. Today, 81 elements are represented in the collection.
Two of them, Thorium and Americium, are not shown in the picture on the
left, which nethertheless does show most of the collection. I don't expect to complete the collection in the sense of 'filling the pokedex' - for of the 118 elements, twenty-two of them are made only in high-grade nuclear reactors and are not available for purchase. In the cases of the heavier elements, their atoms decay into smaller nuclei in a time too short to have a reasonable sample, and many of the heaviest elements are known only from a few atoms. In addition, the purchase of thallium requires a license in the United Kingdom. Which for a metal which you need only touch to get seriously poisoned is probably reasonable. |
| This
page is horizontal! I don't have enough time or willpower to show all
the elements here. But if we look to the right we will find some I find
particularly interesting. |